 May 2021 – Indian Gujarati authorities recommend testing RT LAMP at airports. RT LAMP tests coming soon at Delhi and Mumbai airports. India is another country to finally approve RT LAMP tests for use in COVID-19. pandemic. The test price established by Gujarat government is £40, USD$55, INR 4,070 ! Finally India will get a real time solution !
May 2021 – Indian Gujarati authorities recommend testing RT LAMP at airports. RT LAMP tests coming soon at Delhi and Mumbai airports. India is another country to finally approve RT LAMP tests for use in COVID-19. pandemic. The test price established by Gujarat government is £40, USD$55, INR 4,070 ! Finally India will get a real time solution !
 May 2021 – VIRAQUA Project Team at Bangor University Wales tested our Lamp to test for SARS-CoV-2 in a wastewater study. Out of the three brands that were tested ours was successful and the fastest to produce the results under 15 mins and another rival brand failed to detect. Professor Davey Jones was impressed with our RT-Lamps results and our RT-Lamp has been entered on to the Government Technical register so it’s an approved option available to buy non-clinical testing by departments.
May 2021 – VIRAQUA Project Team at Bangor University Wales tested our Lamp to test for SARS-CoV-2 in a wastewater study. Out of the three brands that were tested ours was successful and the fastest to produce the results under 15 mins and another rival brand failed to detect. Professor Davey Jones was impressed with our RT-Lamps results and our RT-Lamp has been entered on to the Government Technical register so it’s an approved option available to buy non-clinical testing by departments.
A few weeks back DHSC have already concluded a desk top review of our Lamp test and we have passed the data sets, subject to a further independent validation test. Currently they have stock of a rival’s lamp test which was bought a lot earlier and is not being used by them so until that vast stock has been used there will be no progress at governmental level. We have been entered onto the UK Clinical Register so when there is a need then we will be contacted directly by Government bodies.
Across different countries like Poland and Italy and event venues like the European games and at various airports CB-DNA Lamp test is leading the fight against Covid. By providing a fast accurate and practical testing solution!

January 2021 – We are pleased to announce that the CB DNA is the official partner of the European Indoor Athletics Championships 2021 and the Copernicus Cup athletics meeting.
Our RT-Lamp to test all athletes, referees, coaches, volunteers and fans.

#Covid5MinTest
4th November 2020 – Public annoucement in Polish Stock Market
We are proud to announce our distribution agreement with Inno Gene S.A. and their sister research company DNA for UK and Ireland with a target of providing 10m Polish made RT Lamp test to be distributed to Airports, Healthcare, Sports Venues, Industry and Government Bodies. The offical announcement presented below.
Polish Stock Market announcement
FINANCIAL SUPERVISION COMMISSION
Current report no.50 / 2020
Date: 2020-11-04
Issuer's abbreviated name - INNO GENE S.A.
Topic - Conclusion of a distribution agreement by a subsidiary for the SARS-CoV-2 RT-LAMP FAST DETECTION KIT tests
Legal basis - Art. 17 sec. 1 of MAR - confidential information.
Report content:
Management Board of INNO-GENE S.A. _Emitent_ informs that on November 3, 2020, it received information from a subsidiary, i.e. DNA Research Center sp.z o.o. on concluding a distribution and promotion agreement for the SARS-CoV-2 RT-LAMP FAST DETECTION KIT product with London Europe Group Ltd. registered in Great Britain.
The agreement was concluded for an indefinite period and concerns the distribution and promotion of the SARS-CoV-2 RT-LAMP FAST DETECTION KIT test in Great Britain and Ireland. The parties contractually agreed the forecast sales level and the price.
London Europe Group Ltd. _distributor_ has been operating on the British market since 1999 and is the owner of the trademark Virolizer _http: //virolizer.com/contact-us_. Under the trademark Virolizer, the company is heavily selling articles related to the SARS-CoV-2 coronavirus pandemic.
London Europe Group also announces that it is after preliminary talks with management of major airports in Great Britain and Ireland regarding the introduction of the SARS-CoV-2 RT-LAMP FAST DETECTION KIT for passenger screening testing.
The issuer draws attention to the significant value of the contract, estimated at over PLN 50 million.

“Genomic Laboratory of DNA Research Center Poland is a worldclass facility Center, with Proven quality, service and commitment to the scientfic community for many years. Inno Gene S.A. & DNA quality and relaibility of their test is what drove us to join forces in the UK to fight against Covid-19″. DNA is also working on a CovidVax vaccine and we will update you with more developments as they come ”
London Europe Group Ltd – Managing Director – Mr Osman Kaderia
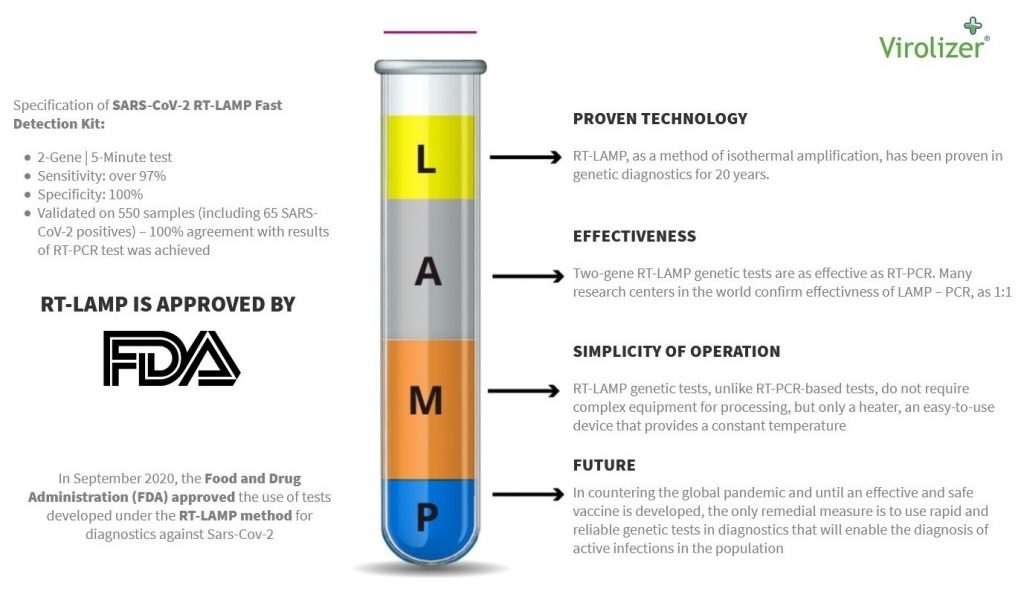
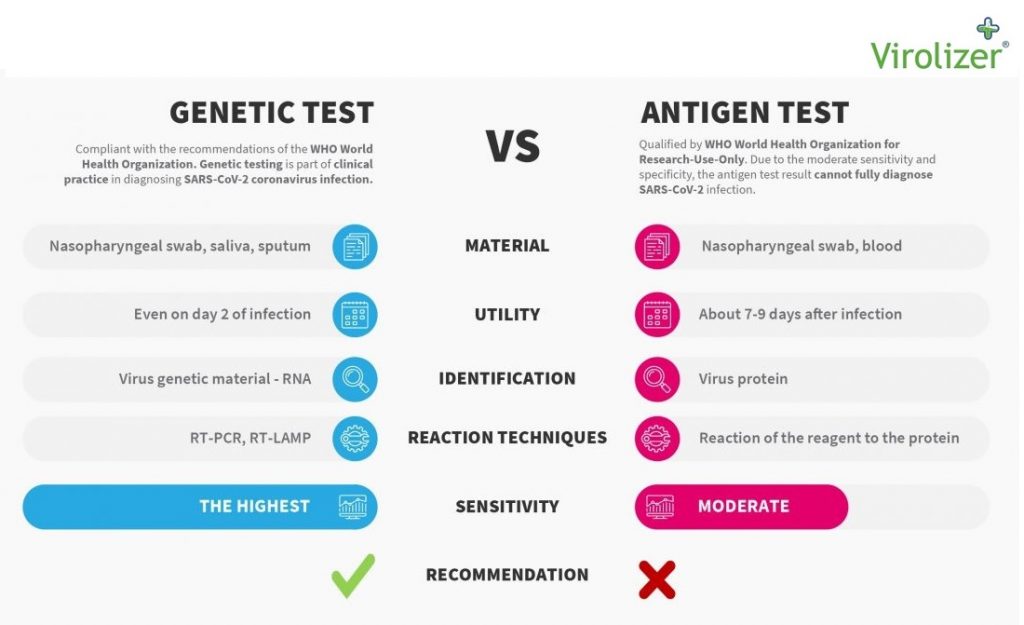
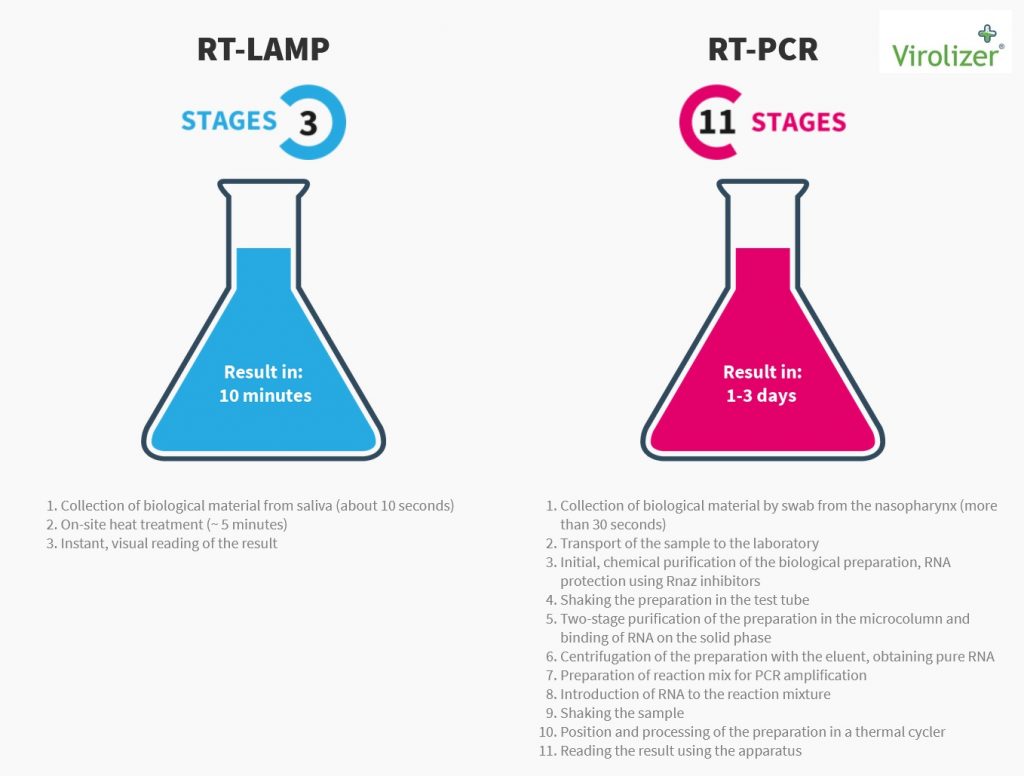


Latest Tests Nov 2020

Inno-gene latest results of live testing show that in 7 cases our unique 2 Gene RT-lamp test detected the Covid19 Virus 2 days earlier then a PCR test in a Polish hospital ! 700 PESA staff at www.Pesa.pl where tested in the last week of November. Average Positive detection time is 2 mins and in some cases we have had results in 48 secs . Now that’s what I call Really Rapid Results ! We can provide this superior product at more than 60% cheaper than our competitors. So I feel that the sooner we are able to offer this to airports and other venue businesses, the sooner we van get back to a form of normality.
*****************************************************************

We are also able to provide a competative made in Europe COVID-19 Antigen Rapid test which can be used by using a swab in the Nasal passage. Accuracy and Reliability is key in these products as peoples lives depend on the outcome. Hence we have selected a European manufacturer rather than and unreliable overseas manufacturer but we can still match the Far East prices. Contact us for Further Information.

Our UKAS registration number is above. We are awaiting a full approval review process.